
Good news! Rui Enni Helps Nasal Inhaler Enterprises Obtain FDA 510 (k) Zero Issue Supplement Again, Certified in 43 Days!This FDA 510 (k) is the 35th zero subsidy case obtained by Ruini's assistance to enterprises!!1、 Introduction to electric nasal suction deviceThe electric nasal cannula is used to intermittently remove nasal secretions and mucus from children (2-12 years old). This device is designed for home environments.Working principle: The electric nasal cannula uses a motor pump to generate negative pressure in the suction system, allowing nasal secretions to flow into the device container. Classification: FDA Class II Medical Devices (Product Code: BTA) 2、 FDA 510 (k) registration experience of electric nasal cannulaDuring the application process for the 510 (k) electric nasal cannula, the Ruini team overcame multiple key challenges with their rich compliance experience and refined project management, ultimately achieving rapid approval with zero replacement. 3、 Registration experience sharing:Product manual: FDA regulations and guidelines are the cornerstone of 510 (k) registration, with detailed and strict requirements. When preparing registration materials, we conducted in-depth research on various relevant standards and guidelines. For example, product manuals must not only comply with electrical safety standards such as ANSI/AAMI ES 60601-1, but also follow FDA guidelines for patient labeling of medical devices. We need to describe the product characteristics and operating methods clearly, accurately, and concisely based on the unique design of the electric nasal cannula, ensuring clear and unambiguous logic. This requires us to cross check every item word for word to ensure compliance with every detail of the regulations.Packaging label: For the outer packaging color box of the product, it is necessary to strictly avoid exaggerating the product's functions and features, and ensure that the promotional content is true and reliable. The FDA attaches great importance to the authenticity of product advertising, and any false or misleading advertising may lead to registration failure. Therefore, when designing the outer packaging, we strictly describe the actual performance and characteristics of the product, without making any exaggerated statements.Performance testing: There is no ready-made industry standard for electric nose pumps abroad, which requires us to develop a testing report that fully complies with the product design and characteristics based on the product's characteristics and rich experience. In terms of performance testing, we cover multiple key indicators such as negative pressure accuracy, noise level, and battery life. Every testing method and requirement has been carefully considered and validated to ensure the accuracy and reliability of the test results.Data authenticity: In order to ensure the authenticity and integrity of the data, we cooperate with third-party laboratories with CNAS and GLP qualifications to conduct various tests that comply with standards.Compliance with standard checklist:Electrical Safety: ANSI/AAMI ES 60601-1Electromagnetic compatibility: IEC 60601-1-2Household environment: IEC 60601-1-11Biocompatibility: ISO 10993-1Software: IEC 62304Lithium battery safety: IEC 62133-2
925
Great news! Assisted Phototherapy Mask Successfully Approved for FDA 510 (k) In the field of medical device regulatory consulting, we continue to write brilliant chapters with our professional strength and unremitting efforts. Today, exciting news came: with the full assistance of our team, the customer's LED phototherapy mask has been successfully approved for FDA 510 (k)! This achievement is not only a powerful proof of the customer's product strength, but also a vivid reflection of our consulting service professionalism and efficiency.1. Introduction to LED Phototherapy MaskLED phototherapy mask is a household medical beauty device mainly used for treating wrinkles on the whole face and mild to moderate acne vulgaris. It cleverly utilizes specific wavelengths of light, including blue light (415nm), red light (630nm), and near-infrared light (830nm), to work in synergy. In acne treatment mode, blue light accurately sterilizes and red light assists in skin repair; In the wrinkle treatment mode, dual light synergistically stimulates collagen production, effectively reducing wrinkles and restoring skin radiance. The product design fully considers ergonomics, with an adjustable headband and a thermal compression function around the eyes (38 ℃ -41 ℃), providing users with a comfortable user experience. This product is quite popular in the markets of China, Europe, and America. It is classified as a Class II medical device in the United States and is marketed through 510 (k) registration. 2. FDA 510 (k) registration experience for LED phototherapy maskIn the early preparation stage of writing the application documents, we conducted comprehensive and in-depth research and analysis of the product with a rigorous attitude and professional spirit. To accurately grasp the core principles of the product, we extensively consulted a large number of authoritative professional literature materials. In the vast sea of information, professional knowledge and rich experience are used to carefully extract concise and clinically based descriptions of working principles, ensuring accurate and scientific expression that can withstand strict scrutiny.In the process of product parameters and testing, we closely integrate FDA guidelines and systematically classify products based on their structural characteristics. Conduct in-depth research on the various parameters that must be provided for this type of product, and carefully study scientific testing methods for different parameters. At the same time, strictly follow the FDA's guidelines on patient labeling and usability to carry out the design and preparation of product instructions. Not only that, but also carefully plan usability testing, covering key testing items such as tag readability, and ultimately issue professional and detailed testing reports.For every content and document that needs to be provided, we uphold an attitude of continuous improvement, from logical structure to written expression, we have repeatedly scrutinized and carefully polished them. Strive to achieve reasonable and coherent content, clear and coherent logic, and easy to understand expression, in order to accurately grasp every detail of the product and provide comprehensive documents, laying a solid foundation for the product to pass FDA review smoothly.3. Apply for key experience sharingBelow is a brief sharing of the problems and experiences encountered during the application process:In the current 510 (k) application process, there are relatively few overall supplementary issues, but there are also some key points. Among them, issues related to biocompatibility, the impact of light on the eyes, and heating modules are particularly prominent.In terms of biocompatibility, as the product is used to treat mild to moderate inflammatory acne, the FDA requires additional biocompatibility testing items related to damaged skin for this purpose. However, based on the FDA's past approval records, similar products do not have consistent practices in this regard. Some similar products have been tested for damaged skin, while others have not undergone this test. Even in the absence of testing on damaged skin using comparable devices with identical expected uses, the FDA still insists on imposing this requirement on our product. This phenomenon fully demonstrates that the FDA's requirements for biocompatibility are becoming increasingly strict, and also reflects the differences in the focus of different auditors. It is worth mentioning that our company, with rich experience and professional prediction, anticipated the possible occurrence of this issue before testing. Therefore, we conducted the test in advance according to the intradermal stimulation standard and informed the customer in advance that there may be a need to supplement the test items according to the damaged skin, making sufficient preparations for the audit.In terms of heating modules, the FDA is highly concerned about their safety, focusing not only on the heating temperature itself, but also on the potential impact of temperature on tissues. This requires us to strictly control the heating module during product design and testing to ensure that it operates within a safe range and avoid potential harm to users.The impact of light on the eyes is also a key focus of the review. The FDA is particularly concerned about whether the product structure can effectively prevent eye damage from light. In addition to optimizing the product structure design, equipping users with goggles that fit the product structure is also a feasible solution to comprehensively ensure the eye safety of users during use.4. Join hands in the future and create more brilliance togetherThe successful approval of the LED phototherapy mask for FDA 510 (k) is another significant achievement of our consulting services. In the future, we will continue to deepen our expertise in medical device regulatory consulting, continuously improve our professional capabilities, and provide high-quality and efficient services to more customers. We look forward to working together with more enterprises to help more innovative medical devices smoothly enter the international market and contribute to the global healthcare industry.Congratulations once again on the successful approval of the FDA 510 (k) for the LED phototherapy mask! We look forward to this product achieving outstanding results in the international market and bringing beauty and health to our users!
751
Rui Enni has helped a company in Beijing obtain CE MDR certification for nutrition pumps, injection pumps, and infusion pumps!Overcoming difficulties and working together to break through:CE MDR is the latest regulation issued by the European Union for medical devices, with clinical safety, technical compliance, and full lifecycle supervision as the core. The certification standards are strict and the process is complex. To assist enterprises in efficiently completing certification, our company has formed a professional team to provide full process consulting services, including product clinical evaluation, CE technical document optimization, and quality management system upgrade (adding MDR QMS).Accurate benchmarking of regulations: Assist enterprises in organizing MDR technical documents, improving risk management (ISO 14971), usability engineering (IEC 62366&IEC60601-1-6), and clinical evaluation evidence;Rapid response and rectification: Provide data supplementation and compliance strategy support for technical questions raised by the notified body (SGS1639);Milestone significanceThe passing of this certification not only demonstrates the technological leadership and international market competitiveness of the enterprise's products, but also sets a benchmark for China's medical device industry to break through the EU's technical barriers. In the future, this series of products will serve European medical institutions, benefit patients worldwide, and lay a solid foundation for enterprises to expand overseas markets.
772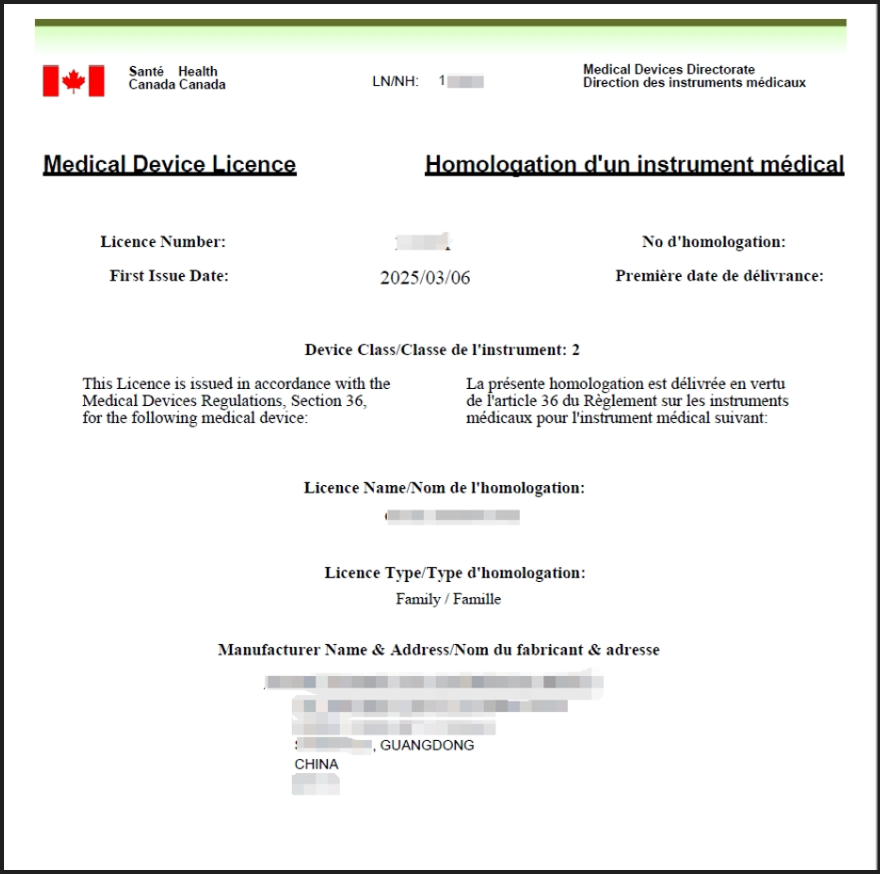
XX Technology, founded in 2006, is a world leading global enterprise in the research and manufacturing of oral health products. It is a national high-tech enterprise and a permanent member of the China Household Appliances Association. We specialize in researching electronic oral care products such as electric toothbrushes, dental irrigators, teeth whitening devices, and ultraviolet disinfectors. Since its establishment, it has continuously invested a large amount of research and development funds, with over 100 R&D engineers and more than 500 patents. It has production bases in Shenzhen, Guangdong, Nanchang, Jiangxi, and Haiphong, Vietnam, with a total area of over 120000 square meters and more than 2600 employees. XX Technology has passed the certification of ISO 9001:2015 Quality Management System, ISO 13485:2016 Medical Device Quality Management System, MDSAP, BSCI, WCA and other relevant industry system standards at home and abroad, and has obtained the GS1 certificate and Dun&Bradstreet Enterprise Code. The product has passed FCC, CE, CB, ETL and other certifications, and is registered with the FDA and the US Food and Drug Administration. With cutting-edge technology, a sound manufacturing system, and cost advantages in overseas factories, XX Technology provides customers with technologically advanced, quality compliant, and cost-effective product services. Since its establishment, it has gained the trust of globally renowned brands and dental clinics, and its products are sold to more than 30 countries and regions.Rui Enni assisted the company in applying for MDL registration in Canada for dental irrigators. The application materials were submitted on February 24, 2025, and the certificate was obtained on March 6, 2025. The certificate was obtained in only 10 days with zero issues and no supplements throughout the process!
894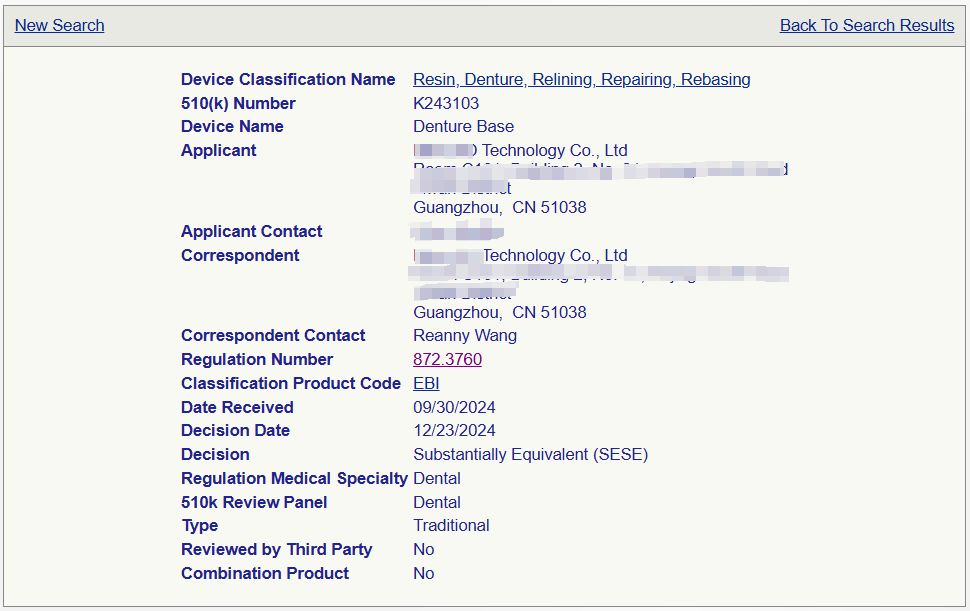
Congratulations to Rui Enni for assisting a customer in Guangzhou in obtaining 510 (k) zero replacement for their 3D additive printed denture base resin, which took 2 and a half months to obtain certification;This application starts with assisting the enterprise in planning various product testing and obtaining Small Business (SBD) certification; And expedite the submission of eStar by the end of fiscal year 2024, successfully obtaining 510 (k) approval with zero defects after 2 and a half months. Saved the enterprise from the increased 510 (k) application fee due to the 2025 fiscal year
1101
Case Company Introduction: XX Technology (L~~~~) is a digital 3D intelligent manufacturer specializing in the research and development of 3D printing equipment, software, and materials, committed to changing the way products are developed and produced. The team has developed high-performance material systems suitable for different industries, relying on independently developed XX series printers and supporting software to provide solutions for innovation and upgrading in industries such as footwear, dentistry, healthcare, consumer, and automotive. They aim to create a new digital manufacturing model and ecological loop that combines customization and mass production.Rui Enni provided MDSAP certification coaching services for the company established in Silicon Valley, USA (USA+Canada+Australia+Japan). We provided remote tutoring for 3 months and successfully obtained the MDSAP certification.
1006Phone
0755-27391220
020-82513196

WeChat customer service

Mini Program
reanny@reanny.com